 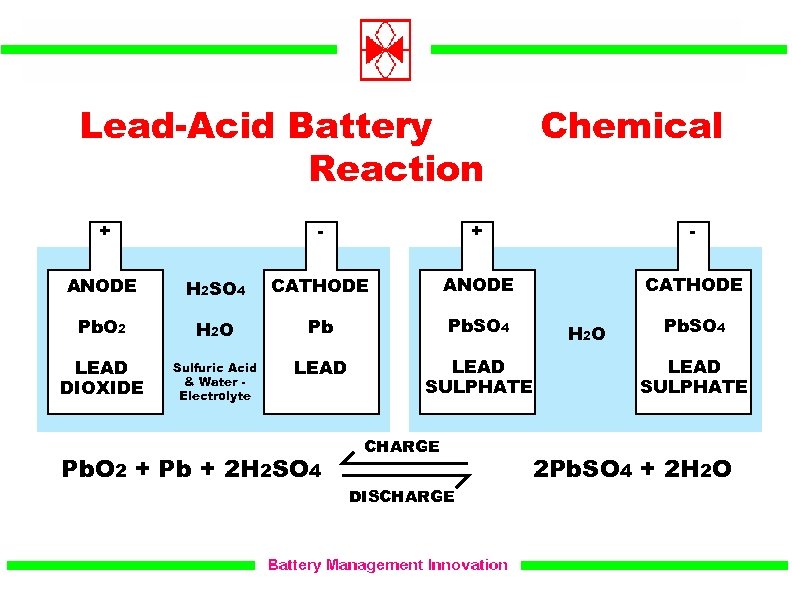
Now if the battery is disconnected from dc source of supply and voltmeter is connected between the electrodes, it will show potential difference between them and on connecting these electrodes, by a wire, current will flow from positive plate to the negative plate through external circuit, i.e., the cell is now capable of supplying electrical energy. Thus during charging lead cathode remains as lead but lead anode gets converted into lead peroxide (PbO 2), chocolate in colour. The oxygen produced attacks the lead anode and forms lead peroxide PbO 2. In the lead-acid battery during charging the cathode reaction is : (A) reduction of Pb2+ to Pb (B) formation of PbSO4 (C) deposition of Pb at the anode (D) formation of Pb02. Each of the negatively charged sulphate ion (SO 4 – –) reaching the anode gives two electrons to it, reacts with water and forms sulphuric acid and oxygen according to chemical reaction SO 4 + H 2O → H 2SO 4 + O. Now if two lead electrodes are immersed in this solution and connected to dc supply mains, the hydrogen ions being positively charged move towards the electrode connected to the negative terminal of the supply mains (i.e., cathode) and SO 4 – – ions being negatively charged move towards the electrode connected to the positive terminal of the supply main (i.e., anode).Įach positive hydrogen ion (H +) reaching the cathode gets one electron from it, becomes atom of hydrogen and escapes out as hydrogen gas in the form of bubbles. When the sulphuric acid is dissolved, its molecules break up into hydrogen positive ions (2H +) and sulphate negative ions (SO 4 – –) and move freely. The lead electrode is in the form of sponge lead.  
In this article we will discuss about the working of lead-acid battery with the help of diagram. The basic anode and cathode materials in a lead acid battery are lead and lead dixodie (PbO2). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
